 
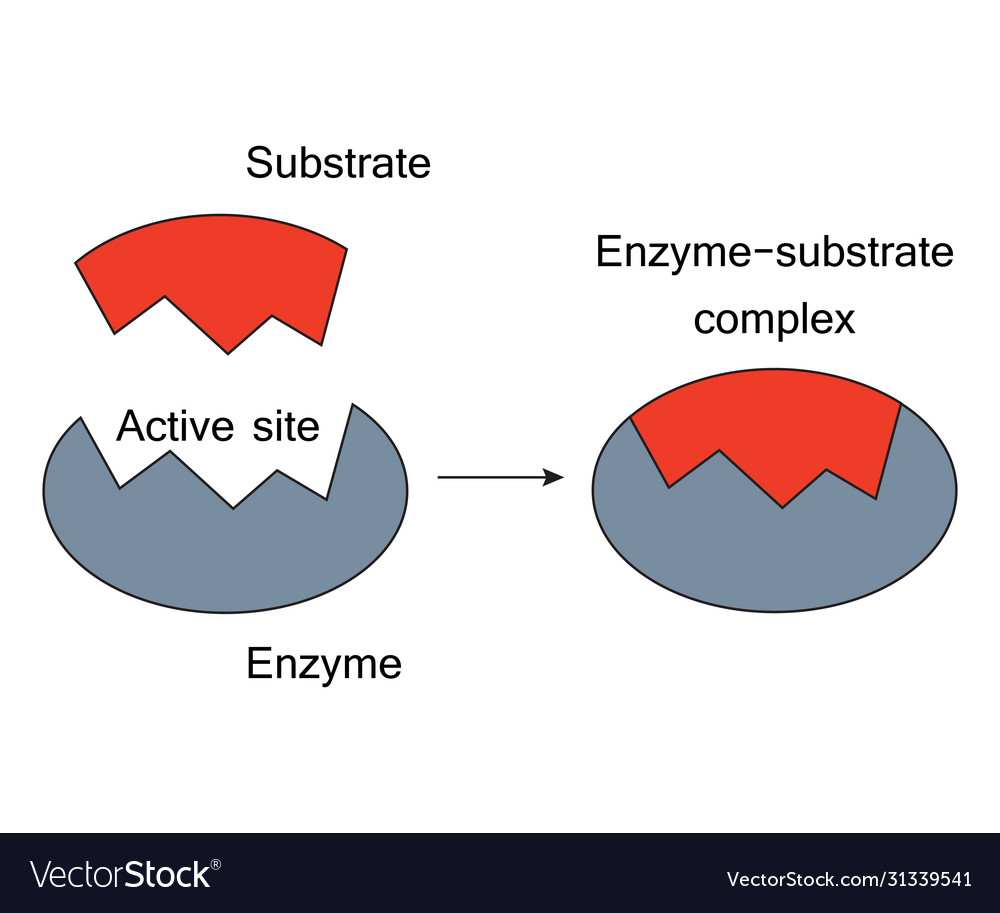
Induced Fit ModelĮvidence from protein chemistry suggests that a small rearrangement of chemical groups occurs in both the enzyme and the substrate molecules when the enzyme substrate complex is formed. The lock and key model for enzyme action accounts for enzyme specificity, because that lack the appropriate shape or are too large or too small cannot be bound to the active site. A specific key can open a particular lock, similarly a substrate having complementary shape and charge to the active site can fit into the active site, and is catalyzed by the enzyme. The active site of enzymes can be compared to key and substrate to a lock. According to this model the substrate has polar (positive and negative) and non-polar (hydrophobic) regions, therefore, it is attracted to and associates with active site which is complementary in both shape and charge distribution. The lock and key model of enzyme action was proposed by Emil Fischer. The substrate is the key that fits exactly the enzyme lock. This idea was originally developed as the lock and key model hypothesis. The specificity 4 enzymes are due to the configuration of the active site. This degree of specificity distinguishes enzymes from other types of catalysts. 
Some enzymes catalyze the transformation of one particular type of substrate molecule or, at most a very restricted group of substrate molecules some catalyzes only one type of chemical change. Lock And Key ModelĮnzymes are highly specific in the reaction the catalyzed. Two models were proposed two explain how Enzymeswork and how active site of an enzyme selects its substrate out of thousands of molecules present in a cell. These two expressions are called Michaelis-Menton constant and Michaelis-Menton equation. They introduced two particularly useful mathematical expressions that for any enzyme relate IS] to V (velocity) and permit quick comparisons of various enzymes catalyzed reactions. Michaelis and Menton are also credited with the first mathematical study of the relationship between substrate concentration and reaction rates. K 3 = rate constant for conversion of S to P. K 2= rate constant for the dissociation of ES In this equation, it is assumed that combination of enzyme and substrate is reversible and 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
